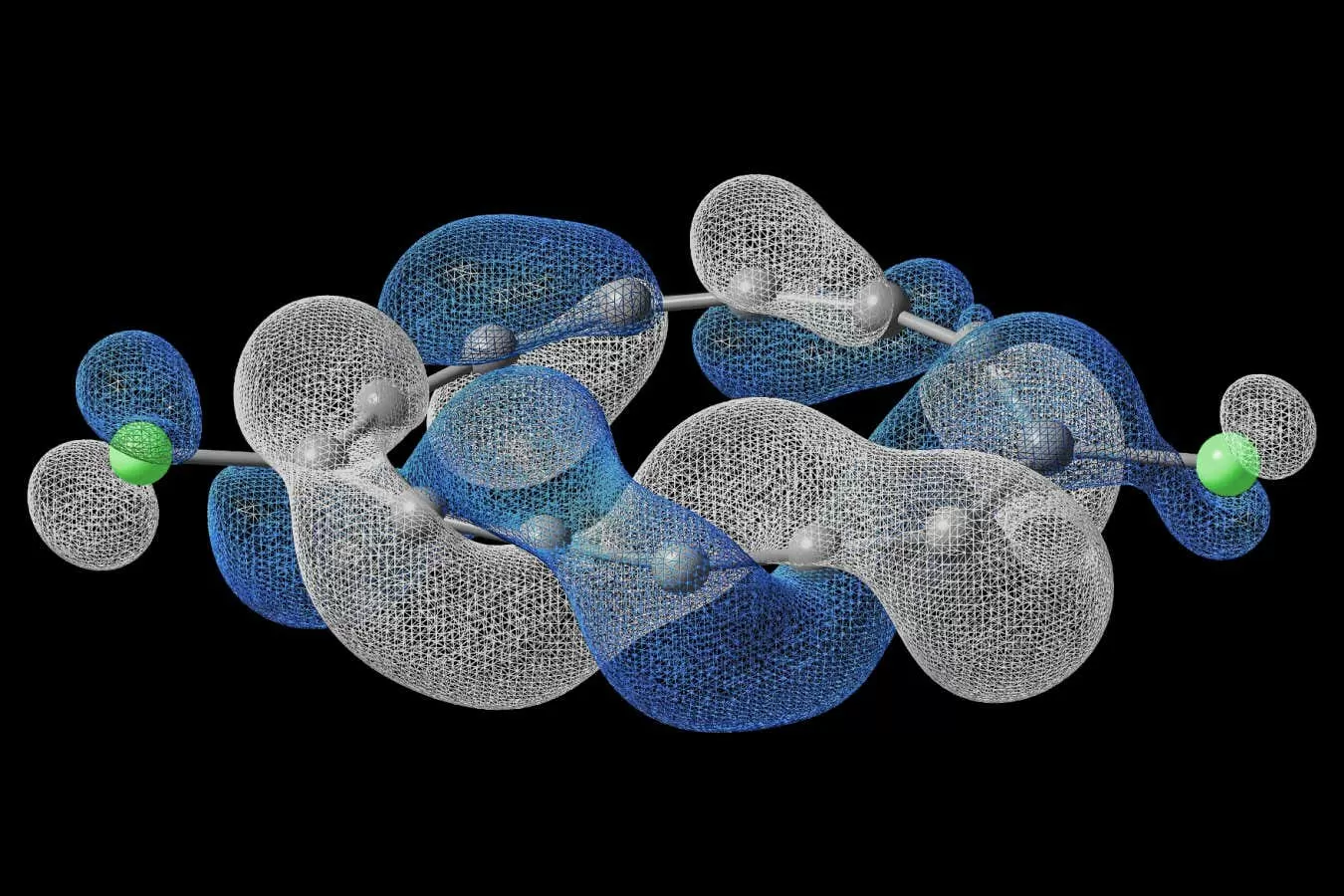
The world of chemistry is full of fascinating structures and compounds that never cease to amaze us. One such compound is a ring of 13 carbon atoms and two chlorine atoms, which has a remarkable molecular structure that is sure to pique your interest. What makes this compound so unique? Well, for starters, it requires you to go around the loop four times to return to your starting position. Let’s dive into the details of this intriguing molecule and explore its properties.
To understand the structure of this compound, we first need to know a little bit about its components. Carbon is a versatile element that can form strong bonds with other elements, making it the building block of life. On the other hand, chlorine is a highly reactive element that is commonly used in disinfectants and bleach. When these two elements come together, they form a compound that is both stable and interesting.
The ring of 13 carbon atoms and two chlorine atoms is known as 1,2-dichlorocyclohexane. It is a cyclic compound, meaning its atoms are arranged in a ring structure. What sets this compound apart is the arrangement of its atoms within the ring. The carbon atoms are bonded to each other in a hexagonal shape, with two chlorine atoms attached to the first and second carbon atoms. This arrangement creates a unique molecular structure that has some surprising properties.
One of the most intriguing properties of this compound is its symmetry. Symmetry is a fundamental concept in chemistry, and it refers to the balance and regularity of a molecule’s structure. In the case of 1,2-dichlorocyclohexane, the symmetry is so perfect that it requires you to go around the loop four times to return to your starting position. This is known as a four-fold rotational symmetry, and it is a rare occurrence in nature.
To understand this concept better, imagine a bicycle wheel with four spokes. If you rotate the wheel by 90 degrees, the spokes will still be in the same position. This is similar to the molecular structure of 1,2-dichlorocyclohexane. The four-fold rotational symmetry means that the compound looks the same from four different angles, making it a truly remarkable molecule.
But why is this molecular structure so significant? Well, for one, it has practical applications in the field of chemistry. The symmetry of this compound makes it an ideal starting material for the synthesis of other compounds. Chemists can use it as a building block to create more complex molecules with specific properties. This is just one example of how the unique structure of 1,2-dichlorocyclohexane can be utilized in the real world.
Moreover, this compound also has implications in the field of biology. Many biological molecules, such as proteins and DNA, have a specific three-dimensional structure that is crucial for their function. The four-fold rotational symmetry of 1,2-dichlorocyclohexane is similar to the symmetry found in these biological molecules. This opens up the possibility of using this compound as a model to study the structure and function of these molecules.
Apart from its practical applications, the molecular structure of 1,2-dichlorocyclohexane is also aesthetically pleasing. The perfect symmetry of the compound is a testament to the beauty and complexity of the natural world. It is a reminder that even the smallest molecules can hold secrets and surprises waiting to be discovered.
In conclusion, the ring of 13 carbon atoms and two chlorine atoms may seem like a simple compound, but its molecular structure is anything but ordinary. The four-fold rotational symmetry of 1,2-dichlorocyclohexane is a testament to its unique properties and potential applications in chemistry and biology. So the next time you come across this compound, remember to appreciate its remarkable structure and the wonders of the chemical world.